Introduction
Gut symbiont restoration therapy is a treatment modality that is developed for autism. We are conducting an academic clinical trial at St. Theresa hospital,5-7-2/775, 3rd LN, Czench Colony, Tulasi Nagar, Sanath Nagar, Hyderabad,Telangana 500038 In this study we will recruit 100 children diagnosed with autism spectrum disorder and treat them with faecal microbiota transfer therapy. Children will be tested for gut microbiome profiles and epigenetic testing before and after treatment along with assessment of severity by childhood autism rating scale.
Goals of the study
- 1) To develop an effective treatment modality for autism
- 2) To understand the cause and pathology of autism
- 3) To develop an antenatal program that can effectively prevent autism
- 4) To develop a serum marker for autism
Investigator :
Dr Chandrashekhar Thodupunuri, is a cardiologist and a MAPS (Medical academy of pediatrics, special needs USA) practitioner. Investigator’s emphasis is on exploring the relationship between gut microbes and human epigenetics. CDFD NGC (center for DNA fingerprinting and diagnostics, National Genomics Core) is supporting this trial for microbiome and epigenetic studies, KSbioclinserve is extending CRO services to this trial. Inductive Quotient analytics is providing data management services for the trial.

Dr. Chandrashekhar Thodupunuri
I am Dr Chandrashekhar Thodupunuri, a cardiologist and a MAPS (Medical academy of paediatrics, special needs USA) practitioner based in Hyderabad. I have realised the need for academic research in autism and kickstarted a research start up “Providence Microbiome Research Centre”. My emphasis is on exploring the relationship between gut microbiome and epigenetics. I believe that the toxins we are ingesting in daily life are disrupting man microbe symbiosis resulting in various diseases; when this symbiotic relationship is affected during pregnancy may lead to autism. N acetylcysteine, a glutathione precursor in the antenatal program, may save children from autism. This possibility is a driving force for us to work in this regard. We actively collaborate with Agenes info omics Pvt ltd and CDFD NGC for microbiome and epigenetic studies, KS Bio Clinserve, Inductive Quotient Analytics for our academic clinical trials.
Practice Places

Resplice Reconnect Center
Address:H no:5-98/RM/6, beside rainbow Maedows villas,near ellenki engineering college,kishtareddypet village,Ameenpur mandal,Sangareddy district,Telangana state,India 502319

St Theresa Hospital
Address: 5-7-2/775, 3rd LN, Czench Colony, Tulasi Nagar, Sanath Nagar, Hyderabad,Telangana 500038
Know More About Autism and GSRT
Autism and Gut Symbiont Restoration Therapy:
Autism spectrum disorder is a highly prevalent neurodevelopmental disorder affecting 88 out of every 10000 children in India. It is commonly diagnosed based on behavioural assessment methods like autism diagnostic observation schedule (ADOS), childhood autism rating scale (CARS), autism treatment evaluation checklist (ATEC) and diagnostic and statistical manual of mental disorders (DSM-5). While the core symptoms of autism are social impairment, deficits in verbal and non-verbal communication, and repetitive movements, the severity is highly variable among individuals, besides suffering from a wide range of comorbid conditions like allergies, constipation, diarrhoea, colic, epilepsy. Despite the lack of an established cause for autism, genetics and epigenetic interactions might be the most probable triggers. Though risperidone and aripiprazole are approved for autism to reduce irritability, no other promising treatment is available except alternative therapies like applied behavioural analysis (ABA), occupational therapy and speech therapy to improve autistic symptoms. Although autism has been growing exponentially since the last three to four decades, no reliable survey reports are available in the Indian population.
| Year | Incidence Rate |
| 1980 | 1 in 10000 |
| 1990 | 1 in 2500 |
| 1995 | 1 in 1000 |
| 2000 | 1 in 150 |
| 2006 | 1 in 150 |
| 2008 | 1 in 88 |
| 2010 | 1 in 68 |
| 2014 | 1 in 59 |
| 2020 | 1 in 54 |
Recent post-mortem brain tissue studies of autistic individuals revealed increased expression of 584 genes. There is a decreased expression of 558 genes in the autistic brain compared to normal healthy controls, and there is differential alternative splicing of 833 genes. Increased or decreased expression refers to the quantity of proteins produced. There is an increased production of 584 neuro proteins and reduced production of 558 neuro proteins in the autistic brain. Eight hundred thirty-three neuroproteins are formed structurally abnormal in autism through a mechanism called differential alternative splicing.
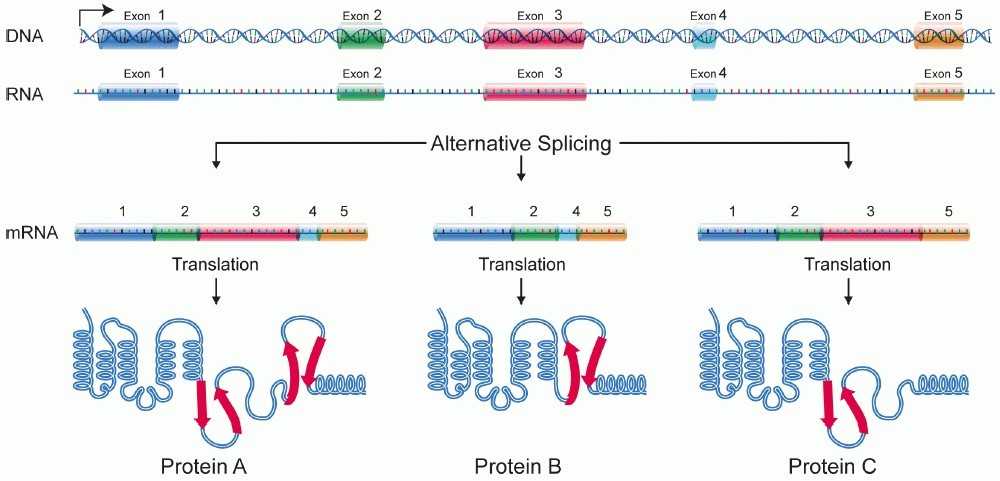
Picture Credit: Wikipedia
As in the picture, If protein A is the desired protein, due to activity dependant down-regulation of neuron-specific exons, protein B or protein C are formed, which are dysfunctional. Thus, differential gene expressions and activity dependant downregulation of neuron-specific exons, i.e. differential alternative splicing produces abnormal quantities of proteins, and structurally abnormal proteins during neuronal development affect the structure and function of the brain.
First, three significant structural abnormalities developed due to epigenetic errors during early brain development in the autistic brain are
1) subependymal nodular dysplasia,
2) heterotopia,
3) multifocal dysplasia.
Second, ongoing epigenetic errors in neuronal function: Around 5 lakh protein turnover per minute takes place at synaptic junctions. Therefore, any abnormal protein synthesis patterns or structurally abnormal protein (isoform) formation will affect nerve conduction or nerve functioning. This phenomenon may explain sensory issues and GI motility issues in autistic individuals.
There are two possible causes for epigenetic errors in the autistic brain.
1) Genetics
2) Gene-Environment Interactions.
Epigenetic errors in autism are similar to those in Rett’s syndrome, with some differences. Gene mutations like Mecp2 in Retts syndrome are expected for autism. Simmons foundation autism research initiative (SFARI) and simons powering autism research identified around 1000 genes associated with autism. None of them had a prevalence of more than 1% in total autism diagnoses.
Gene-environment interactions are another possible cause of epigenetic errors. The onset of epigenetic errors in the autistic brain occurs during intrauterine life;

Picture Credit: Illustration by Benjamin Arthur for NPR
hence the gene-environment interaction must be abnormal during intrauterine life itself.
There are two major environmental factors in the uterus.
1) Cord Blood
2) Uterine Microbiome
- Cord Blood: The environmental working group and Rachel network did a cord blood study and revealed around 232 toxicants, potentially neurotoxic during early brain development.
- Uterine Microbiome: until recently, the uterus was thought to be sterile. Recent studies demonstrated that there are 18 taxa of microbes that are species specific and evolutionarily related to human beings; microbes like micrococcus luteus and Bacteroides thetaiotaomicron etc. can influence human gene expression patterns through which they can actively involve in fetal development, and fetal immune system development etc. it is hypothesized that these microbes are species specific and evolutionarily related and act as pioneering microbes in fetal intestines and direct the development of human-compatible ecosystem
Microorganisms produce inducer molecules or signaling molecules to communicate with each other in the colony. These inducer molecules influence their gene expression and other microbial genes in the territory. This genomic interaction is the language of communication among microbes. This mechanism is also called a quorum sensing mechanism. Similar mechanisms exist between the human body and gut microbes; hence, a human-compatible gut ecosystem is necessary.
Tomova et al. studied gut microbiome profiles in autistic children and demonstrated the presence of a phylogenetically different ecosystem that has nitriliruptor, youngiibacter, methanomicrobiales and bilophila as essential organisms.
Gil Sharon et al. transplanted stool from autistic individuals into a pair of germfree mice, and their offspring were studied. The germ-free mice that received a faecal transplant from autistic individuals produced autistic offspring. Their brains showed differential gene expression patterns and differential alternative splicing patterns similar to autistic human being’s brains, which is evidence of the genomic interactions between microbes and host (human being or mice or any multicellular organism)
In a mice study, pregnant mice were exposed to bisphenol A produced behaviourally abnormal mice. Similar studies with various pesticides exposures to pregnant mice had abnormally behaving mice.
Hence, It is hypothesized that toxins like bisphenol A and pesticides promote phylogenetically different and human incompatible microbiome drives these epigenetic errors that affect brain structure formation and function.
According to us, there may be two possibilities that could be responsible for epigenetic errors.
- Cord blood toxins act as neurotoxicants and drive abnormal neuroprotein formation during early brain development.
- Cord blood toxins might kill or prevent pioneering microbes, i.e. uterine microbiome, from colonizing in the fetal intestine. Microbes capable of surviving toxic environments may be colonizing first and driving a human incompatible ecosystem that triggers epigenetic errors during brain development resulting in abnormal brain structure formation. This human incompatible ecosystem drives epigenetic errors throughout an individual’s life, resulting in dysfunction of the developed part of the Brain and trigger sensory issues, GI issues, and abnormal immune responses throughout life. It is estimated that around 5000 genes all over the body showed differential function in autism, affecting many biological processes.
Gut symbiont restoration therapy is designed in expectation of reversing epigenetic errors by restoring the evolutionarily related symbiotic microbes by Faecal microbiota transfer. Although reversal of ongoing epigenetic errors may help improve neuronal functions like sensory processing defects and GI motility, aberrant immune responses etc., consequences of epigenetic mistakes of the past, like structural abnormalities of the brain due to abnormal neuronal protein formation during brain development, may or may not reverse. For E.g. Reelin production is reduced by 40% in the autistic brain, causing neuronal migration defects during brain development. Though reelin production is restored after FMT, structural abnormalities may or may not be repaired, depending on neuroplasticity. However, structural abnormalities secondary to neuronal migration and differentiation defects may reverse if human and symbiotic microbe relation is restored in the active neuroplasticity period (first few years of life).
Faecal microbiota transplantation (FMT) can be administered through oral capsules, nasogastric tubes, endoscopy, colonoscopy, enemas, etc. FMT can be a potential modality of treatment for various gastrointestinal conditions and neurological disorders etc. Donor health, stool quality of donor, and recipient’s gut environment are critical for the success of FMT. Gut symbiont restoration therapy is designed with a strategic bowel cleanse and multiple sittings of faecal microbiota transfer therapy to ensure the eradication of human-incompatible ecosystem and engraftment of the human-compatible ecosystem in children with autism. The success of faecal microbiota transfer can be assessed by Investigating the microbiome profiles before and after treatment using 16s rRNA sequencing. In addition, the efficacy of FMT can be investigated by evaluating changes in epigenetic changes in the blood. Successful colonization of micrococcus luteus and other microbes of uterine microbiome and their lineage may be an indicator of success of FMT
A safe donor can be a healthy, playful, joyful child, preferably a sibling or family member or close relatives or well-known people whose health profile is very much known. A healthy child possesses all essential taxa of microbes, which constitutes the evolutionarily related primary microbiota needed for optimal health and development. If a recipient receives all the vital bacteria, an excellent diversity can be achieved by nutritional interventions.
In a breakthrough study published in April 2019, microbiota transfer therapy showed a significant improvement in children with autism, including 44% of children recovering with no residual autism over two years, which inspired us to work in this regard.
Microbiota is a potentially modifiable factor that can modulate epigenetics of the host, through which it can affect metabolism and immunity etc. Hence microbiota from a healthy donor can reverse abnormal gene expression and restore normal development, provided the transplant is done in window periods with intact neuroplasticity.
POSSIBLE OUTCOMES AFTER FAECAL MICROBIOTA TRANSFER THERAPY
GENETICS
Chromodomain Helicase DNA protein 8, 15q11.2-12.3 duplication syndrome, autism risk genes like cellular adhesion molecules (CAMs) and synaptic scaffold proteins -neurexin, neuroligin, contactin, cadherin gene families, members of general immunoglobulin CAM superfamily, SH3 and multiple ankyrin repeat domain proteins (SHANK) and SNPs etc
GENE-ENVIRONMENT INTERACTION
Some Gut microbes can interact with human genes in a similar way they interact with each other using quorum sensing mechanisms. In addition, microbes produce inducer or signalling molecules that can influence human gene expressions
IRREVERSIBLE
REVERSIBLE
Gross epigenetic errors and protein quality control
STRUCTURAL CHANGES
FUNCTIONAL CHANGES
Proteins are building blocks in the human body. During brain structure development, abnormal quantities of protein production and structurally abnormal protein formation result in
- Subependymal nodular dysplasia,
- Heterotopia
- Multifocal dysplasia. Fundamental mechanisms affected are
- Accelerated neurogenesis
- Impaired neuronal migration
- Poor neuronal differentiation
- Altered neuroimmune mechanisms.
THESE CHANGES ARE REVERSIBLE TILL CERTAIN AGE IF NON-GENETIC
The human brain collects information from the surroundings through sense organs and processes that information and responds to external stimuli. This process involves signal transmission in the form of neurotransmitters, which involves high protein turnover, abnormal splicing patterns and differential
protein formation during this process results in dysfunction leading to sensory issues, speech, communication deficits and repetitive behaviours, echolalia etc., a similar mechanism in the enteric nervous system result in GI issues. There are developed and underdeveloped areas in the autistic brain,
developed part is also dysfunctional in autism due to abnormal RNA processing and differential protein synthesis.-REVERSIBLE IF NON-GENETIC.